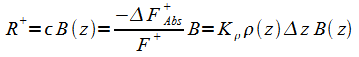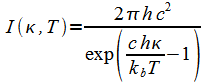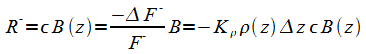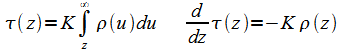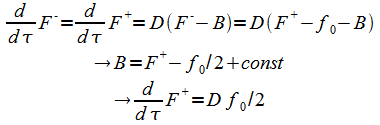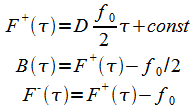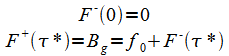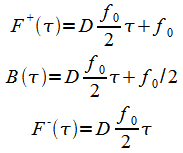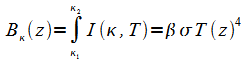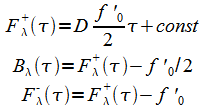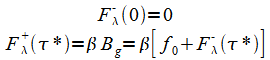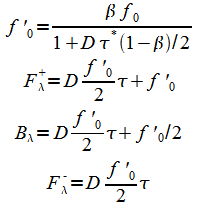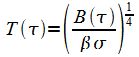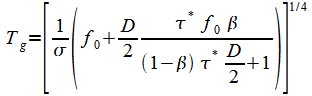Narrow Band Schwarzschild Radiation Balance Equations
A Simple Accurate Climate Model
How to Calculate Absorption from HITRAN data
Why is Controlled Nuclear Fusion so Difficult?
Reaction Rate,Cross Section & Activation Energy
Introduction
The radiation balance equations of
K Schwarzschild describe how heat absorbing gasses in the
atmosphere of a planet will warm the planet by trapping the heat
re-radiated from the surface of the planet. The original theory
proposed back in 1906 grossly overestimates the amount of warming.
However a simple modification3 where the heat
absorption is limited to a narrow band of radiation frequencies
greatly improves the accuracy of the theory.
The first section
of this derives the Schwarzschild equations in detail. Next they
are modified for a narrow absorption bands; finally the modified
equations are applied to the atmospheres of Mars and Earth. The
calculations show a 25% increase of CO2 in the atmosphere of Earth
will produce a surface temperature increase of about a tenth
degree Celsius
Schwarzschild Radiation Balance Equations—Original wide band version
Figure 1 depicts radiation flowing thru a section of atmosphere of thickness Dz.
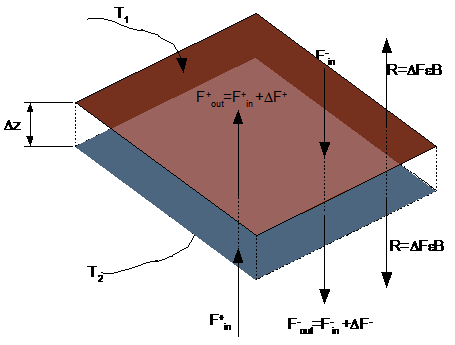
Fig.
1: Energy flow through a layer of atmosphere
The radiation flow is categorized into upward and downward flows F+ and F-.Upward flowing radiation F+in is entering the portion of absorbing-emitting material. A portion DF=Fout-Fin is absorbed by the material; The amount of radiation energy absorbed is proportional to the distance,Dz (positive going upwards) and is also proportional to the the radiation intensity itself:
|
|
(1) |
The
absorbed radiation
![]() is
re-emitted as R+
in
accordance with Kirchhoff's law; The total upward flowing
radiation on the outbound side of the block is comprised of the
incident radiation less that absorbed plus the radiation R that is
emitted from within the block. The intensity of the emitted
radiation is:
is
re-emitted as R+
in
accordance with Kirchhoff's law; The total upward flowing
radiation on the outbound side of the block is comprised of the
incident radiation less that absorbed plus the radiation R that is
emitted from within the block. The intensity of the emitted
radiation is:
|
|
(2) |
Where DF
from (1) is substituted into (2), and the fraction of energy
absorbed
![]() is
equated to the emissivity according to Kirchhoff law. Use a minus
sign because DF is negative for
absorption.
is
equated to the emissivity according to Kirchhoff law. Use a minus
sign because DF is negative for
absorption.
Generally, B(z) is related to Planck's function. For wide spectral bandwidth situations in gray-body type atmospheres, B(z) is taken as Planck's function integrated over all frequencies—the Stefan-Boltzmann function:
|
|
(3) |
If the frequency k is measured in wave number units, then Planck's function I(k,T) is:
|
|
(4) |
R+ in (2) adds to F+ in (1):
|
|
(5) |
For the downward flowing energy we have:
|
|
(6) |
Sign changes in (6) because Dz from F-in to F-out is negative. The downward version of (2) becomes:
|
|
(7) |
Substitute (7) into (6) to get the downward version of (5):
|
|
(8) |
The differences become differentials in the usual way in (5) and (8)to get:
|
|
(9) |
|
|
(10) |
Up to now, only the energy flow in two directions—up and down have been considered. Obviously, energy can flow in any direction. For the situation of a planet atmosphere, where the radiation is locally isotropic, it's possible to integrate equations (9) and (10) over the possible directions and that leads to the simple modification:
|
|
(11) |
|
|
(12) |
The constant D is generally taken to be 3/2 though other values are used. This is a standard part of the theory and is discussed in the book by Goody and Yung for example. It turns out in the that the final value of temperature is not very sensitive to the value of D in the narrow band modification.
Change of Variable z to Optical Depth t
As long as Kr is constant so that the absorption is only a function of the distance z, (11) (12) can be rewritten in a solvable way through the introduction of the optical distance variable t defined as:
|
|
(13) |
Variable t is dimensionless and physically represents the logarithm of the relative radiation attenuation over the distance z. Since r decreases with increasing z, t also increases with increasing height. Writing (11) (12)in terms of t:
|
|
(14) |
|
|
(15) |
The energy fluxes F± are not independent. Their sum equals the externally applied radiation source f0(the Sun in other words):
|
|
(16) |
So with the help of (16) we can eliminate F- to get:
|
|
(17) |
And the general solution of(14) and(15) is
|
|
(18) |
The constants are determined from the boundary conditions:
|
|
(19) |
With t* being the mapping at zero height:
|
|
(20) |
From definition (20) together with boundary condition (19) into (18):
|
|
(21) |
Temperature is determined from (3).
Schwarzschild Radiation Balance Equations--Narrow Band Absorption
These equations with some modifications (Weaver and Ramanathan 3) can be applied to portions of the spectrum provided that Kirchhoff's law holds on a frequency by frequency basis. We consider the case where absorption takes place over a limited range of frequencies. The emitted power (3) becomes:
|
|
(22) |
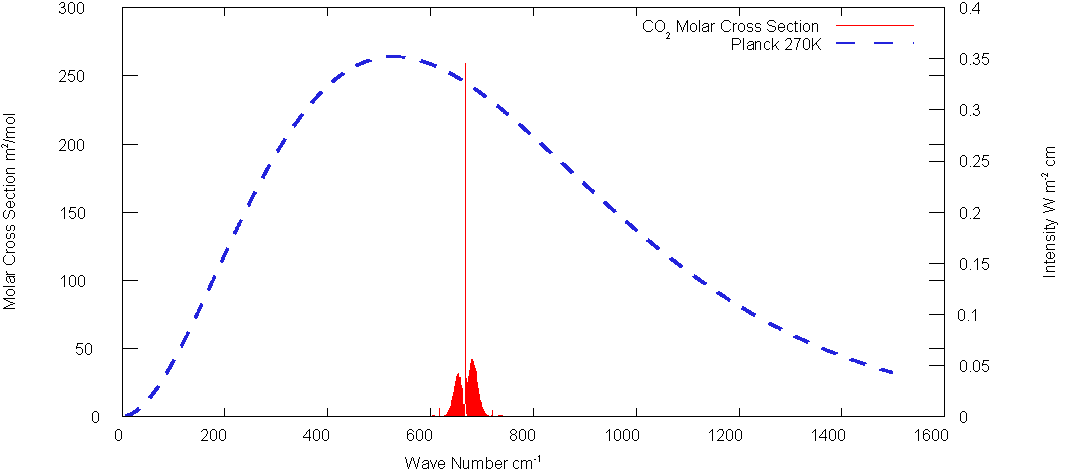
Fig.
2: CO2 Spectrum
eq. (22) defines b which is a function of temperature owing to the changing shape of Planck's distribution with temperature. As a simplifying assumption, take b constant.
The energy balance equation (16) is modified—the difference between upward and downward energy flux is no longer the total energy input from the Sun, but a fraction thereof to be determined from the boundary conditions.
|
|
(23) |
Generally, f''0 is temperature dependent, but again simplify to a constant.
Equations (18) are likewise modified:
|
|
(24) |
The boundary conditions are:
|
|
(25) |
Note that f0 is not primed in (25) because we assume the ground absorbs all the radiation( 95% for earth though it's easy to allow a an emissivity <1.); only the fraction b is available for atmosphere absorption. From (24) and (25):
|
|
(26) |
From Eq.(22), the temperature as a function of optical depth is:
|
|
(27) |
To obtain the ground temperature use t* in (27). to get:
|
|
(28) |
Applications
Carbon Dioxide parameters:
There are actually two relevant absorption bands for CO2 which are evident the following plot of Fig. 2 on a log scale(The band at about 2500 cm1- is totally outside the 270K spectrum).
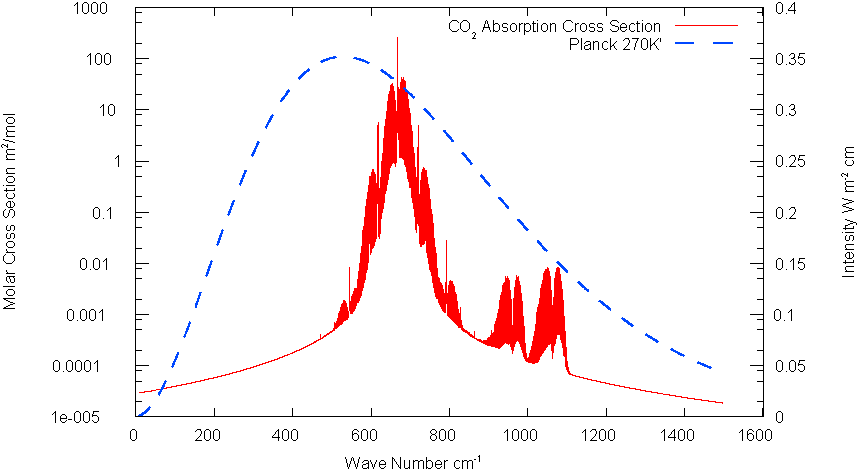
Fig.
3: CO2 Spectrum Log Scale
The specific cross section K which defines t eq(13) is applied only to the absorbed band of frequencies. Value calculated from HITRAN data is 5.25 m2/mol for a band 200 cm -1 wide centered about 667cm-1.(Half width 100 cm-1)The parameter b of eq. (22) is about .108. for this bandwidth. The band centered at about 1050 cm-1 has specific cross sec tion of only about 7x10-4 cm2/mole.
Calculate optical depth
For simplicity, take temperature as constant Tavg in the hydrostatic relation:
|
|
(29) |
Rm is molar gas constant; g is gravity acceleration,MCO2 is molecular weight of Carbon Dioxide.
The optical thickness (20) at ground level works out to be:
|
|
(30) |
Where Kk is the specific molar cross section applied to the band of absorbed frequencies;p0 is the surface pressure.
Example: Mars
Mars is a good place to test the theory as its atmosphere is 95% carbon dioxide. The surface pressure is only about 600 Pa—less than .6% that of Earth,but it would stack to a height of over 80 meters if squeezed to STP conditions. By contrast, Earth's CO2 component at 380 ppmv would occupy a height less than 2 meters under the same conditions. So if one compares the respective CO2 components, the Mars atmosphere is actually very thick relative to that of Earth. Calculation parameters are shown in table 1. Two values of D are used. They bracket the various accepted values and have little effect on the surface temperature. The bands at 667cm-1 and 1050 cm-1 warm the planet about 6K and 4K. Add the two to the black body temperature to get a surface temperature of about 222K.
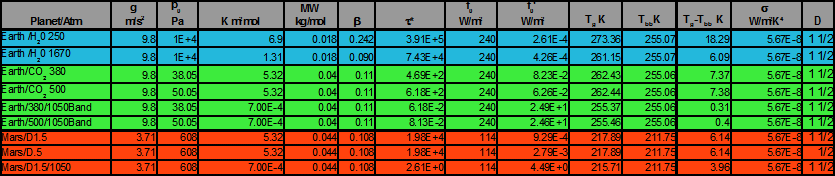
Table
1: Calculation Results
Earth

Table 1: Calculation Results
At 380 ppmv(300K), the calculations shows that carbon dioxide warms the atmosphere about 7.37. This band s saturated; if the concentration were raised to 500ppmv the warming would only increased to 7.38K. The band at 1050cm-1 contributes about .3K warming and that would increase to about .4K at 500ppmv.
Table 1 also shows the calculated effect of water vapor. There are two areas of absorption; the main band is centered at roughly 250 cm-1 and contributes nearly 24 K warming. The other band though much wider, is located on the tail of the Planck spectrum and contributes only about 6 K warming.
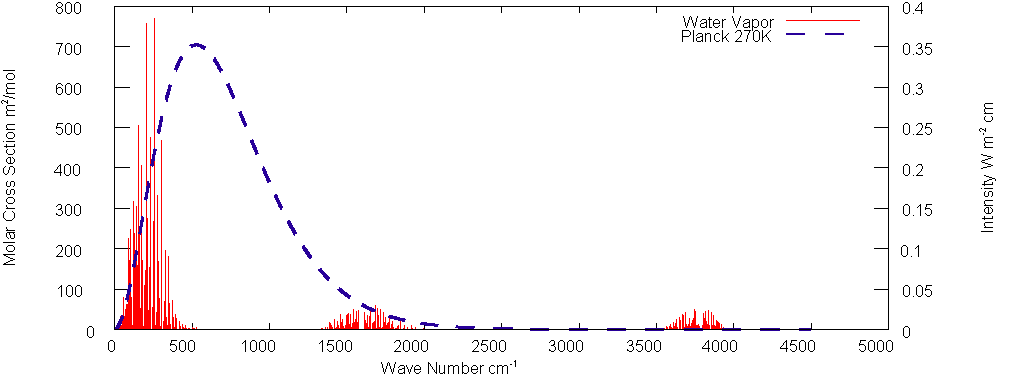
Fig.
4: Water Vapor Absorption
Atmospheric Temperatures
Write the optical thickness parameter t as a function of height using the simplified hydrostatic relation 29:
|
|
(31) |
Using only the water vapor data in eq. 27 and 31:
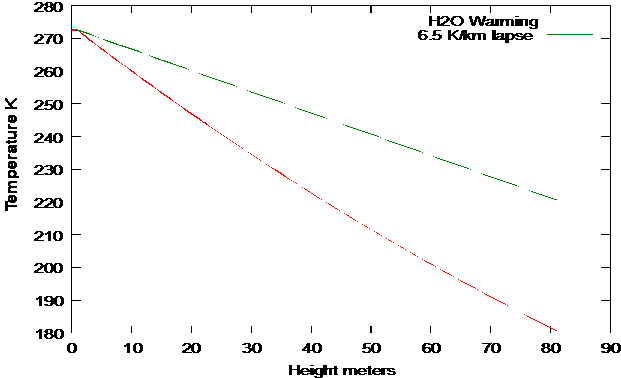
Fig.
5: Predicted and Actual Lapse rate
We see that the actual temperature lapse
rate is significantly less than the theory. But that is to be
expected. The adiabatic cooling rate induces instability in the
atmosphere and that's what makes a changing climate. The details
of the process are beyond the scope of this theory.
Conclusions
This simple modification of the classic Schwarzschild equations to allow for limited absorption bandwidth of the heat absorbing gasses in an atmosphere produces excellent agreement with observations on both Earth and Mars. The calculations also show that a 25% increase of CO2 in the atmosphere will produce a temperature increase of about .1 °C.
References
1. Goody ,R, M. and Y.L. Yung, Atmospheric Radiation:Theoretical Basis,2nd ed. Oxford University Press, New York, 1989.
2. Pierrehumbert, R.T. Principles of Planetary Climate
http://geosci.uchicago.edu/~rtp1/
3. Weaver, C.P. Weaver and V. Ramanathan, Deductions from a simple climate model: Factors governing surface temperature and atmosphereic thermal structure, J. of Geophysical Research v.100 no. D6 pages 11585-11591